Friday Report: Issue 64
By: John Roberts, Adele Groyer, Matt Fletcher and Dan Ryan
COVID-19 is still one of the hottest topics for scientific papers and articles. The COVID-19 Actuaries Response Group provides a bi-weekly Friday update with a summary of key papers, articles and data.
Vaccines
Moderna announces trial results for COVID vaccines in children under age 6 (link)
In a trial of around 6,700 under 6-year olds, effectiveness against infection of between 38% (age 2-5) and 44% (6 to 24 months) was observed. These relatively low levels are cited as consistent with that seen in adults with regard to Omicron infection. For these age groups a quarter dose (25µg) was administered, and no untoward side effects were observed.
Moderna is now moving ahead with submissions for global regulatory approval for its vaccine for these age groups, along with approval from the FDA in the US for 6- to 11-year olds.
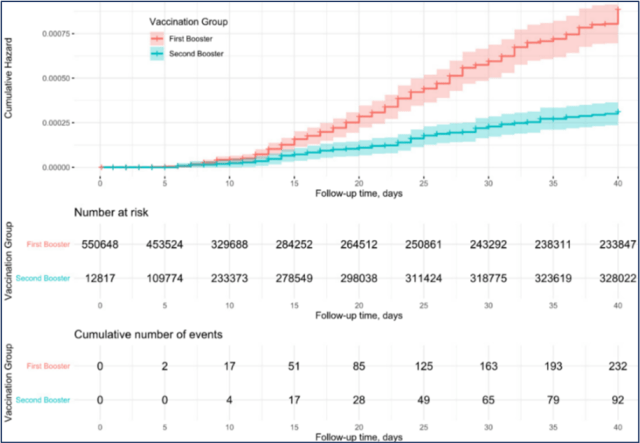
Study from Israel confirms 2nd booster effectiveness (link)
A study of over half a million adults aged over 60, of whom 329,000 had received a second booster, shows a dramatic reduction in deaths after the second booster. The hazard ratio is quoted at 0.22 (95% CI: 0.17, 0.28), meaning a 78% effectiveness against death, compared with those who have only had the first booster. The study also quotes hazard ratios for age bands within this group, and this shows that the booster is equally effective across all ages, when the wider confidence intervals are taken into consideration.
Follow-up time was limited to 40 days, so the longevity of the additional protection is not assessed in this study.
A second study (link) also from Israel, but with a smaller sample population of around 100,000 participants quotes vaccine effectiveness against severe disease of 86% (CI 73% – 93%), with little waning (as compared with three doses only). However, against infection it notes a reduced effectiveness of 64%, further reducing to 29% by the tenth week of the study.
With the Spring Booster campaign now under way in the UK for the over-75s or immunocompromised, these studies are a useful reminder of the benefit of this additional jab.
Infection- and vaccine-induced immunity similar for Omicron BA.1 and BA.2 (link)
This preprint is an investigation into protection offered by five forms of immunity against symptomatic infection, hospitalisation and death, for Omicron BA.1 and BA.2 infection. It’s based on analysis of the Omicron waves in Qatar, starting 19 December 2021 and peaking in mid-January 2022.
The forms of immunity investigated were:
- Prior infection with pre-Omicron variants
- Two or three doses of Pfizer or Moderna vaccines
- Hybrid protection with vaccination and prior infection
Hybrid immunity with prior infection and a recent booster was found to have the strongest protection against both variants, and no discernible differences were found in protection against BA.1 compared with BA.2

Variants
BA.2 Growth (link)
The latest Variant Technical Briefing confirms that the rapid rise of BA.2 has continued, and by late March was around 94% of all cases. The Briefing notes a growth advantage over BA.1 of around 75% since mid-February, although thankfully there is no evidence of greater risk of hospitalisation. Nevertheless, the increased transmissibility results in increased numbers of admissions, as we have seen over recent weeks.

Medical
New studies on effectiveness of candidate COVID-19 treatments
On 30 March, the New England Journal of Medicine published two studies on the effectiveness of COVID-19 treatments.
In one study (link), convalescent or control plasma was administered by transfusion to participants with COVID-19 in the US. Hospitalisation occurred in 17 of 592 participants (3%) who received convalescent plasma and 37 of 589 participants (6%) who received control plasma. In participants, most of whom were unvaccinated, the administration of convalescent plasma within 9 days after the onset of symptoms was associated with a relative risk reduction of 54%.
In terms of safety, 34 adverse events were noted in the convalescent-plasma group and 55 in the control-plasma group.
In another study (link), as part of the TOGETHER trial, 3,515 patients from clinics in Brazil were randomly assigned to receive Ivermectin (679 patients) or placebo (679). The primary outcome investigated was an emergency department visit or hospitalisation within 28 days of randomisation. Overall, 100 patients (14.7%) in the Ivermectin group had a primary-outcome event, as compared with 111 (16.3%) in the placebo group (relative risk, 0.90; CI, 0.70 to 1.16). From this the authors conclude that treatment with Ivermectin did not result in a lower risk of the primary endpoint studied.
In terms of safety, 163 adverse events were noted in the Ivemectin arm and 201 in the control arm.
Research on damage from microthrombi
Early in the pandemic researchers flagged the possibility that microthrombi were a key contributor to the systemic damage that we see in COVID-19. Thankfully, we already have a number of drugs that have anti-thrombotic activity, and these fall into two groups. Anti-coagulants drugs, or blood thinners, act by inhibiting clotting factors in the blood. Anti-platelets drugs act by stopping platelets forming clumps in the blood.
In May 2020, doctors at Mount Sinai Hospital, USA started trials on anti-coagulants that might counteract the creation of these microthrombi (link). Then in August 2021, three large international clinical trials published in the New England Journal of Medicine confirmed that anti-coagulant treatments could reduce the need for organ support for COVID-19 patients outside ICU (link).
Now in March 2022, the BHF Data Science Centre’s CVD-COVID-UK Consortium (link) has published results from a very large cohort (link) that examined the benefits of pre-existing treatments with both classes of anti-thrombotics for patients with a prior history of atrial fibrillation and/or at high risk of stroke. The cohort consisted of nearly 1 million patients drawn from 56 million electronic health records across England; 3.8% of the cohort were admitted to hospital with COVID-19 and 2.2% died over the period up to May 2021. 88% of the cohort were already being treated with anti-thrombotic treatments, and pre-existing treatment was associated with 8% lower mortality.
Closer investigations of the cohort identified that those receiving anti-coagulant treatments had a 7% lower mortality rate than those receiving anti-platelet treatments, and that there was no difference between direct oral anti-coagulants and warfarin. The figure below shows that use of anti-thrombotic treatments varies by co-morbidities and demographic factors. It also shows that the benefits in mortality were not due to a lower likelihood of being admitted to hospital.

Organ impairment and Long COVID (link)
This preprint is a one-year prospective longitudinal cohort study into Long COVID. The study recruited 536 Long COVID patients, most of whom had not been hospitalised following a COVID infection. MRI scans of lungs, heart, kidney, liver, pancreas and spleen were carried out at six months after COVID infection for the 536 patients, and followed up for 331 individuals at twelve months. A smaller healthy control group was also scanned.
The study found that the burden of symptoms reduced over time (from a median of 10 symptoms six months post-infection down to 3 at twelve months), but that single organ impairment persisted in 59% of individuals at 12 months after infection. The authors note that they do not have history and imaging of the individuals pre-pandemic, making it difficult to determine whether the impairment was caused by COVID-19 or Long COVID.

T-Cell Reponses to infection remain durable after 12 months (link)
A study of 141 patients (out of an initial enrolment of over 1,000) who had moderate to critical initial COVID disease, assessed both antibody and T-cell responses over a 12-month period.
The study found that whilst antibody responses were stable at both 6 and 12 months after initial infection for those under 60, these were reduced in respect of three variants, including Beta and Delta. In contrast, T-cell responses were maintained, notably for those for whom no antibody response was detected. The authors conclude that cross-reactive SARS-CoV-2-specific T-cell responses could be particularly important in the protection against severe disease caused by variants of concern whereas neutralising antibody responses seem to reduce over time. 
Data
Mortality risk for younger people post vaccination (link)
This report from ONS provides an analysis of mortality following COVID-19 vaccination and excess mortality during the pandemic in people under the age of 29 in England.
ONS found no evidence of a change in the number of deaths either from cardiac-related conditions or from any cause following COVID-19 vaccination in people aged 12-29. There were more deaths registered in people aged 15-29 in 2021 than the average over 2015-2019, but ONS note that the impact of the pandemic on mortality in younger people is still unclear because deaths from external causes take a significant amount of time to investigate – this is especially true for 2020 where the operation of coroners’ courts was disrupted by lockdowns.
Early data suggests that the number of deaths in 2020 increased for some causes, particularly suicides in young females and accidental poisonings in young males.
ONS Infection Study (link)
Since our last update two weeks ago prevalence in England is estimated to have increased from 4.9% to 7.6%, which is its highest figure recorded. Wales and Scotland have also seen sharp increases, with Scotland recording 9.0% last week, the highest ever recorded.
In contrast to the situation we’ve seen for a lot of the pandemic, there’s a much closer spread by age range now, with infection levels climbing rapidly in the older age groups. As an example the 70+ cohort level is modelled to have increased by 74% between the 12th and 26th March.


ONS Antibody Study (link)
This fortnightly update continues to show very high levels of antibodies at all ages, with around 99% for all adult age groups. We noted in our last report that ONS suggests an increase in the threshold used may be necessary to provide the equivalent level of protection against Omicron as the current threshold did against Delta. As such, these sustained high figures may not be representative of waning immunity against Omicron.
We will therefore pause regular updates on these results until there is something meaningful to report.
New UKHSA study into vaccine booster effectiveness against hospitalisation (link)
A study by UKHSA released as a pre-print has shown that waning of vaccine effectiveness against hospitalisation may be overstated when considering all hospitalisations associated with a positive COVID-19 test. UKHSA considered vaccine effectiveness under various COVID hospitalisation definitions and varying by length of stay.
Far less waning is seen for more severe types of hospitalisations that specifically involve respiratory issues as a primary diagnosis. Defining hospitalisations in this way overcomes distortions from incidental Covid hospitalisations which are now more common in the presence of Omicron.
The study controlled for multiple factors associated with risk of hospitalisation including age, health status and wider determinants of health.
Vaccine effectivness observed against the different types of hospitalisation is summarised below.
| Ages 18 to 64 | Ages 65+ | |||
| Peak effectiveness post-booster | 15+ weeks post booster | Peak effectiveness post-booster | 15+ weeks post booster | |
| All Covid hospitalisations | 82% | 54% | 92% | 77% |
| Admissions with a respiratory code lasting 2+ days | 91% | 67% | 91% | 85% |
| Admissions requiring oxygen, ventilation or intensive care | 97% | 76% | 96% | 87% |
The apparent greater waning among younger people should be treated with caution as the report shows that confidence intervals are particularly wide at the later durations post-booster in this age group where boosters were rolled out more recently than at older ages.
Other
News from California (link)
It’s inevitable that we can cover only a fraction of the COVID news and research in our bulletin, and so we were pleased when Dr Ruth Ann Crystal got in touch to tell us of a similar publication, albeit with a different format, which she publishes, with the collaboration of alumni, students and Professors of Stanford University.
The weekly update is also published on a Friday, although given the time difference, will probably also be available to UK readers on Saturday morning.
And Finally …
Masking Musicians (link)
We recently noticed the ultimate in COVID protection for musicians – a face mask that allows you to continue playing your instrument. They are claimed to be suitable for all instruments, though why you would need this particular feature for stringed or percussion instruments we’re not quite sure!

The masks do seem to miss the point that when playing most wind or brass instruments, the breath is forced through the instrument, so they would appear to have minimal benefit. Maybe a mask that fits over the bell of a French horn might be more effective?
1 April 2022





Pingback: So how do we feel about Covid now? | AgeWage: Making your money work as hard as you do