Friday Report: Issue 54
By: Adele Groyer, John Roberts and Dan Ryan
COVID-19 Actuaries Response Group – Learn. Share. Educate. Influence.
COVID-19 is still one of the hottest topics for scientific papers and articles. The COVID‑19 Actuaries Response Group provides a regular Friday update with a summary of key papers and articles.
Vaccines
Booster and 12 to 15 Roll-out
The programme to give Priority Groups 1 to 9 booster doses continues, with heavy criticism of the government of the pace of the roll-out over the last two weeks, both in the media and in parliament (including for full disclosure by one of the authors). As shown below, progress has steadily increased, but remains below those becoming newly eligible. After a brief dip below 2m in early November, the “required run rate” will settle at around 2.2m for the next month. link
As of 29th Oct, the total given a booster in England is 5.9m, approximately half of those now eligible, and just over a quarter of the estimated 22m that will become eligible in due course.

Meanwhile, the latest data on the roll-out of first doses to 12 to 15 year olds shows steady progress, though not at the rate that we have seen for other age groups. These figures are to Sunday, so won’t yet reflect the change in policy to permit “walk-ins”, (as opposed to restricting vaccination to the school hosted service), which only started this week.

Approval Nearer for Pfizer vaccine for 5 to 11 year olds in USA (link)
In a meeting earlier this week independent advisors for the FDA recommended approval of emergency use of the Pfizer vaccine for younger children. The near unanimous vote now paves the way for a roll-out to around 30m children next month, with the FDA expected to give formal approval, in advance of consideration by the CDC on November 2nd.
Neurological complications COVID-19 vaccination and SARS-CoV-2 infection
Identification of rare adverse events is important for ongoing risk-benefit evaluation of vaccines and for informing post-vaccine clinical practice. In this study (link) the authors investigated rare neurological complications associated with COVID-19 infection and vaccinations.
The study design was a self-controlled case series which is commonly used for the evaluation of vaccine safety. Individuals act as their own controls but the case and control events and exposures are partitioned by time. In this study baseline exposure is determined 29 days or more before as well as after the events of interest – vaccination with a first dose of ChAdOx1nCoV-19 or BNT162b2 COVID-19 vaccines or a SARS-CoV-2 positive test. The study population consisted of the 32.5m people in England who received their first dose of COVID-19 vaccine between 1 December 2020 and 31 May 2021. In this population of vaccinated people, 2m had a SARS-CoV-2-positive test, of which 1.8m had their SARS-CoV-2 test prior to vaccination.
With ChAdOx1nCoV-19 there was an increased risk of Guillain–Barré syndrome and Bell’s palsy at 15–21 days after vaccination.
With BNT162b2 there was an increased risk of haemorrhagic stroke at 15-21 days after vaccination.
However there was a substantially higher risk of all neurological outcomes in the 28 days after a positive SARS-CoV-2 test including Guillain–Barré syndrome. Overall the study estimated 38 excess cases of Guillain–Barré syndrome per 10 million people receiving ChAdOx1nCoV-19 and 145 excess cases per 10 million people after a positive SARS-CoV-2 test.
The authors conclude that, although they found an increased risk of neurological complications in those who received COVID-19 vaccines, the risk of these complications is even greater following a positive SARS-CoV-2 test.

SARS-CoV-2 Neutralization with Pfizer–BioNTech Vaccine Dose 3
As part of an ongoing global, randomised, placebo-controlled trial of the BNT162b2, a third dose was administered 7.9 to 8.8 months after dose 2 to 11 participants 18 to 55 years of age and to 12 participants 65 to 85 years of age from U.S. sites. Serum specimens were obtained before dose 1, at 7 days and 1 month after dose 2, and before and 7 days and 1 month after dose 3. The observed neutralisation titres against Wild Type, Beta and Delta variants were reported in the New England Journal of Medicine (link).
The third dose resulted in higher neutralisation geometric mean titers (GMTs) after 1 month than was observed 1 month after dose 2. Additionally neutralisation GMTs decreased from 7 days to 1 month after dose 2 but increased from 7 days to 1 month after dose 3. A similar pattern of broader neutralisation (i.e., against variant strains) and higher GMTs after dose 3 was seen in assays with the delta variant spike protein on a wild-type genetic background. The geometric mean ratio of neutralisation GMTs (delta variant to wild type) 1 month after dose 3 was 0.85 in younger adults and 0.92 in older adults.
This suggests that a booster dose of BNT162b2 administered 7 to 9 months after the primary two-dose series could prolong protection and further increase the breadth of protection.

Infection after Vaccination (link)
With such a high proportion of the UK population vaccinated, and despite this very high infection rates, a relevant question is what impact does infection subsequent to vaccination have on our immune system.
A relatively small study based in Massachusetts contrasted antibody titers at a median time of 6 months after vaccination in those subsequently infected or not. Vaccinated infected individuals had markedly higher binding and neutralizing antibody responses as well as higher cellular immune responses compared with vaccinated uninfected individuals, suggesting that breakthrough infections triggered robust anamnestic immune responses.
One notable point is that the infected individuals have a significantly younger age profile. However, the authors observe that there was no particular correlation between age and level of response amongst the participants.

SARS-CoV-2 transmission by vaccination status
A study led by Imperial College London and the UK Health Security Agency (link) and (link) aimed to investigate transmission and viral load kinetics in vaccinated and unvaccinated individuals with mild delta variant infection in the community.
Between 13 September 2020, and 15 September 2021, 602 community contacts of 471 UK COVID-19 index cases were recruited into the study.
Outcomes studied were the secondary attack rate (SAR) in household contacts stratified by contact and index case vaccination status. For the viral load kinetics analysis, peak viral load, viral growth rate, and viral decline rate were studied.
The SAR in household contacts exposed to the delta variant was 25% (95% CI 18–33) for fully vaccinated individuals compared with 38% (24–53) in unvaccinated household contacts.
The median time between second vaccine dose and study recruitment in fully vaccinated contacts was longer for infected individuals (median 101 days [inter-quartile range 74–120]) than for uninfected individuals (64 days [32–97], p=0·001). This suggests that the risk of infection increased within 3 months of receiving a second vaccine dose, due to waning protective immunity. The authors point to this finding as important evidence of the need for vaccinated people to get a booster shot as soon as they are eligible for one.
The SAR among household contacts exposed to fully vaccinated index cases was similar to household contacts exposed to unvaccinated index cases (25% [95% CI 15–35] for vaccinated vs 23% [15–31] for unvaccinated)
Peak viral load did not differ by vaccination status or variant type. Fully vaccinated individuals with delta variant infection had a faster mean rate of viral load decline.
The authors conclude that vaccination reduces the risk of delta variant infection and accelerates viral clearance. Nonetheless, fully vaccinated individuals with breakthrough infections have peak viral load similar to unvaccinated cases and can efficiently transmit infection in household settings.
Clinical and medical news
Announcement on ritonavir anti-viral treatment
In the recent government press briefing intended to update on progress with booster vaccines, Secretary of State for Health and Social Care, Sajid Javid, announced that the UK government was purchasing 480,000 courses of molnupiravir and 250,000 courses of PF-07321332/ritonavir “ritonavir”, all anti-viral treatments that had been originally flagged in April for further investigation. We covered recent clinical trials for these anti-viral treatments in our 1st October bulletin (link) . In the press conference, Sajid Javid emphasised that use of these purchased treatments was only subject to MHRA authorisation.
There are three Phase 2/3 trials for ritonavir that are at different stages of recruitment, examining the benefits for post-exposure prophylaxis (EPIC-PEP), treatment for non-hospitalised standard risk patients with COVID (EPIC-SR) and treatment for non-hospitalised high risk patients with COVID (EPIC-HR).
The last of these trials (EPIC-HR) was started on July 16 (link) and is continuing to recruit 3,000 participants in the USA. This trial has a primary completion date of December 9 and a study completion date of April 21, 2022. As we outlined in our previous bulletin, a Phase 1 trial of ritonavir has completed and demonstrated that the treatments are safe and well-tolerated, but as this involved healthy individuals it would not be expected to provide any data on efficacy against COVID in humans. The only public efficacy data is in respect of in-vitro studies that state that ritonavir has “potent anti-viral activity against SARS-CoV-2.”
Whilst any news regarding potential new therapies to combat the virus is welcome, the timing of such an announcement and the possible expectation it gave the public in terms of the immediacy of use of the drugs may be questioned, given that MHRA is unlikely to grant approval until successful completion of the Phase 2/3 clinical trial, most likely after the winter. In addition, it is not clear whether the RECOVERY trial is expected to evaluate the treatment prior to authorisation, as has been the case for other drugs.
Long Covid in German children, adolescents and adults
In this pre-print study (link) the authors investigated long-term health sequelae using healthcare data of 45% of the German population from January 2019 to December 2020. 157,134 individuals (11,950 children/adolescents and 145,184 adults) with confirmed COVID-19 were matched in a 1:5 ratio to controls on age, sex and prevalent medical conditions. COVID-19 and control cohorts were followed for incident morbidity outcomes documented at least three months after the date of COVID-19 diagnosis.
Overall, 96 pre-defined outcomes were considered. For all health outcomes combined, incidence rates in the COVID-19 cohort were significantly higher than those in the control cohort:-
- Children/adolescents: IRR=1.30, 95%-CI=[1.25-1.35]
- Adults: IRR=1.33, 95%-CI=[1.31-1.34]
Incidence rates in children/adolescents were consistently lower than those in adults.
Specific outcomes with the highest IRR and an incidence rate of at least 1/100 person-years in the COVID-19 cohort in children and adolescents included:
- malaise/fatigue/exhaustion IRR=2.28, 95%-CI=[1.71-3.06]
- cough IRR=1.74, 95%-CI=[1.48-2.04]
- throat/chest pain IRR=1.72, 95%-CI=[1.39-2.12]
In adults the outcomes with the highest IRRs included:
- dysgeusia (altered sense of taste) IRR=6.69, 95%-CI=[5.88-7.60]
- fever IRR=3.33, 95%-CI=[3.01-3.68]
- dyspnea (shortness of breath) IRR=2.88, 95%-CI=[2.74-3.02]
The authors conclude that the study indicates substantial new-onset post COVID-19 morbidity. Further investigation is required to assess the persistence and long-term health impact of post COVID-19 conditions, especially in children and adolescents.
Sotrovimab for early treatment of COVID-19
Sotrovimab, formerly known as VIR-7831, is an engineered human monoclonal antibody that neutralizes SARS-CoV-2 and multiple other sarbecoviruses. (Sarbecovirus is a subgenus of betacoronavirus comprising the coronaviruses linked to severe acute respiratory syndrome including SARS-CoV -1 and SARS-CoV-2) This study (link) funded by Vir Biotechnology and GlaxoSmithKline reports the interim results of an ongoing, multicentre, double-blind phase 3 trial.
583 non-hospitalised patients with symptomatic Covid-19 but within 5 days of symptom onset and with at least one risk factor for disease progression received a single infusion of sotrovimab at a dose of 500 mg or placebo.
3 patients (1%) in the sotrovimab group, as compared with 21 patients (7%) in the placebo group, had disease progression leading to hospitalisation or death. The treatment is associated with a relative risk reduction of 85%; 97.24% confidence interval, 44 to 96 and P=0.002
Modelling
With the release of new SPI-M models last week (link), it is inevitable that all the focus will be on those, with much less given to outcomes in comparison with previous versions. However, we’ve been tracking hospital admissions compared with a previous model, and for completeness show the latest position here.
We can see that the predicted peaks, with the exception of the Warwick “green” version have not materialised (note we don’t show Imperial’s curve, as it is off the scale). However, there has been a much more sustained level than expected.

In terms of the new models, which we will start tracking from now on, one interesting point to note is that they understate the current position, being at around 600 admissions per day, whereas we are currently averaging 830 per day.
Data
UKHSA Variants Technical Briefing (link)
The latest Technical Briefing includes an in depth analysis on the Delta sub-variant AY.4.2. In terms of transmissibility the data suggests a 19% advantage over “original Delta”, which is in line with previous thinking of the relative attack rates (and which intuitively must be the case if the sub-variant is growing as a proportion of overall cases). The briefing notes that the growth in AY.4.2 has not been consistent throughout the regions, either in terms of speed or timing.
However, there is yet no evidence to suggest that the sub-variant is more vaccine resistant, nor that it results in worse clinical outcomes.

ONS Antibody Study (link)
After a gap of over a month due to unspecified quality issues, the ONS’ antibody study has returned, with levels across the UK remaining just above 90%. Of more interest is what is happening at the older age groups, and here we can now clearly see evidence of waning antibody prevalence.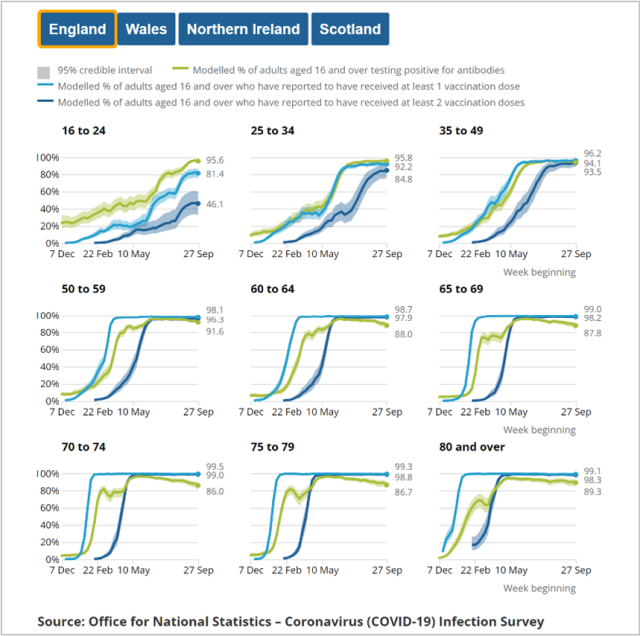
As has been noted before, it is helpful to plot the proportion of those not registering antibodies, and this clearly shows an increasing trend, typically from a low of around 4% up to between 10% and 12% by the fourth month. This level of increase is reasonably consistent with data we have previously reported on from PHE (link) which similarly shows an increase in the “efficacy gap” as duration following the second dose lengthens.

ONS Infection Survey (link)
The last two weeks have seen substantial increases in infection levels in England and Wales with the former increasing from 1.63% to 2.02%. Elsewhere the favourable trends seen in Scotland and Northern Ireland appear to have reversed, dramatically so it would appear in Northern Ireland, although some of this may be due to statistical volatility given the wide confidence intervals for the smallest of the four nations.
Regionally the South West has seen the strongest growth in the last month, and is now the region with the highest prevalence. By age, much of the increase has been driven by school age children, with school years 7 to 11 now put at a prevalence of 9%. Older ages are growing too, although at a much lower level, so the scaling forced by the high prevalence is school children hides this in the graphs.
‘R’ Estimate (link)
The estimate for England has risen in the last fortnight from (0.9 to 1.1) to (1.1 to 1.3). Last week saw no regional estimates, because of the Immensa testing lab false negative issue. This week regional estimates are resumed, except in the South West, which continue to be paused given the uncertainties surrounding testing there. Nevertheless, the UKHSA expresses confidence that R is above 1 in the region.

And Finally
Planning on some halloween decorations this weekend? Hopefully you won’t atract the attention of the police unlike this unlucky chap! At least the officer concerned had a sense of humour…

Have a good weekend, whatever your plans.




I followed up one of the actuaries’ links to SAGE minutes (which was instructive and seemed to be good common sense), but was surprised to find so many redactions amongst the names of the Observers and Government attendees.
I struggle to think of a reason why such heavy redaction would be required. Any ideas, Henry?